  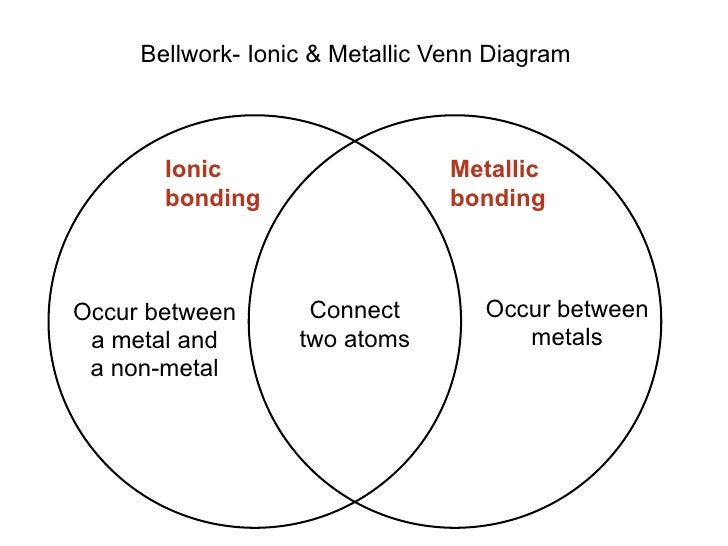 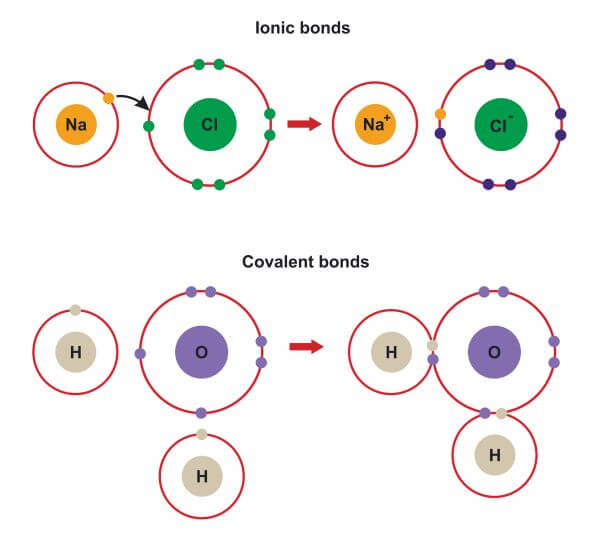
Maybe, on may admit that Boron atom prefers to stay covalent rather than to loose three electrons. Lewis structures for covalent bonding use lines to represent the sharing of electrons. Why ionic solutions conduct an electrical current while. MOLECULAR or COVALENT compound, while sodium chloride (table salt) is an IONIC compound. For example, one point near $\ce$ remains covalent, and it is hard to justify. How to tell whether a substance will dissolve to form an ionic solution or a molecular solution. The difference between ionic and molecular compounds is that ionic compounds are composed of positive and negative ions held together by ionic bonds. Then consider the outer electron layers : put enough points around each symbol to get a reasonable Lewis structure. Ionic crystals - The ionic crystal structure consists of alternating positively-charged cations and negatively-charged anions (see figure below). In principle, when studying a structure like HCl, CH3COOH, or any other compound, the first thing to do is drawing the symbol of the atoms in a reasonable geometry on a sheet of paper. MOLECULAR COMPOUNDSIONIC COMPOUNDSThe interaction of molecular compounds occurs between neutral atoms or nonmetals.The interaction of ionic compounds occurs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
